 I shall attempt to outline one of these methods*.Īll molecules are assumed to be spherical and of equal size and situated on a lattice with co-ordination number z. Entropy of Mixing Last updated Ideal Systems Ideal Gas Processes A gas will always flow into a newly available volume and does so because its molecules are rapidly bouncing off one another and hitting the walls of their container, readily moving into a new allowable space. The increase in entropy in this case comes from the irreversibility of the process. Note that the nal state (unlike the initial state) is one of di usive (or chemical) equilibrium. It comes about from a combination of the entropy of expansion and the distinguishability of the particles. This deviation gives rise to excess heat capacities of mixing and in consequence to excess vibrational entropies of mixing. The behavior of a system becomes more complicated when the system contains two or more types of particles (aka mixtures). This increase in entropy is called the entropy of mixing. In solid solutions, the configurational entropy is unequal to zero and the heat capacity versus composition behaviour generally deviates from a linear relationship, especially at low temperatures of 50 K. Free Energy of Mixing Calculating G m i x should be no more difficult than calculating S m i x. The diffusion of initially separated gases result in an. Consequences: the energy of intermolecular interactions is the same for all the molecules, and the entropy is reduced because of the indistinguishability of particles. The entropy change for a system undergoing isothermal mixing is always positive. When the initial pressures of and are equal and the "remove barrier" is selected, which corresponds to mixing at constant pressure, the entropy of mixing is, where and are the mole fractions of and in the final mixture.The calculation of the mixing energy is extremely complicated and only approximate solutions have been obtained as far as I am aware. Spontaneous processes net increase in entropy increase in randomness of distribution (mixing). and x B are the mole fractions of the two components, and the enthalpy of mixing is zero. For an ideal solution the entropy of mixing is assumed to be. The Thermodynamics of Regular and Ideal Solutions Ideal and Regular Solutions. The entropy of mixing polymers is so small that ev en an extremely small unfavorable enthalpy of mixing can prevent mixing. Liquids and Solutions II: 2nd Year Michaelmas Term. 2 His argument for excess-entropy scaling was based on the quasiuniversality of simple liquids traditionally explained by reference to the hard-sphere system based van der Waals picture of liquids. The entropy of the gas in the two-container system can be easily calculated, but if the equation is not extensive, the entropy would not be 2 S. Rosenfeld reported computer simulation results for simple model liquids of point particles like the well-known Lennard-Jones (LJ) system. No macroscopic changes occur, as the system is in equilibrium. As the pressures increase, the color becomes more intense. Entropy of mixing, polymers As N 00, Xc 0 and the chains become immiscible at all temperatures (see Figure 31.9). Now a door in the container wall is opened to allow the gas particles to mix between the containers. Gas is colored red and gas is colored blue, and when the gases mix, different shades of purple result, depending on the ratio of moles of each species. The total entropy change is the sum of the entropy changes of each gas. For "compress right", if the partial pressure of a gas does not change, its entropy does not change, even when mixed with another gas. When the partial pressure decreases, entropy increases. 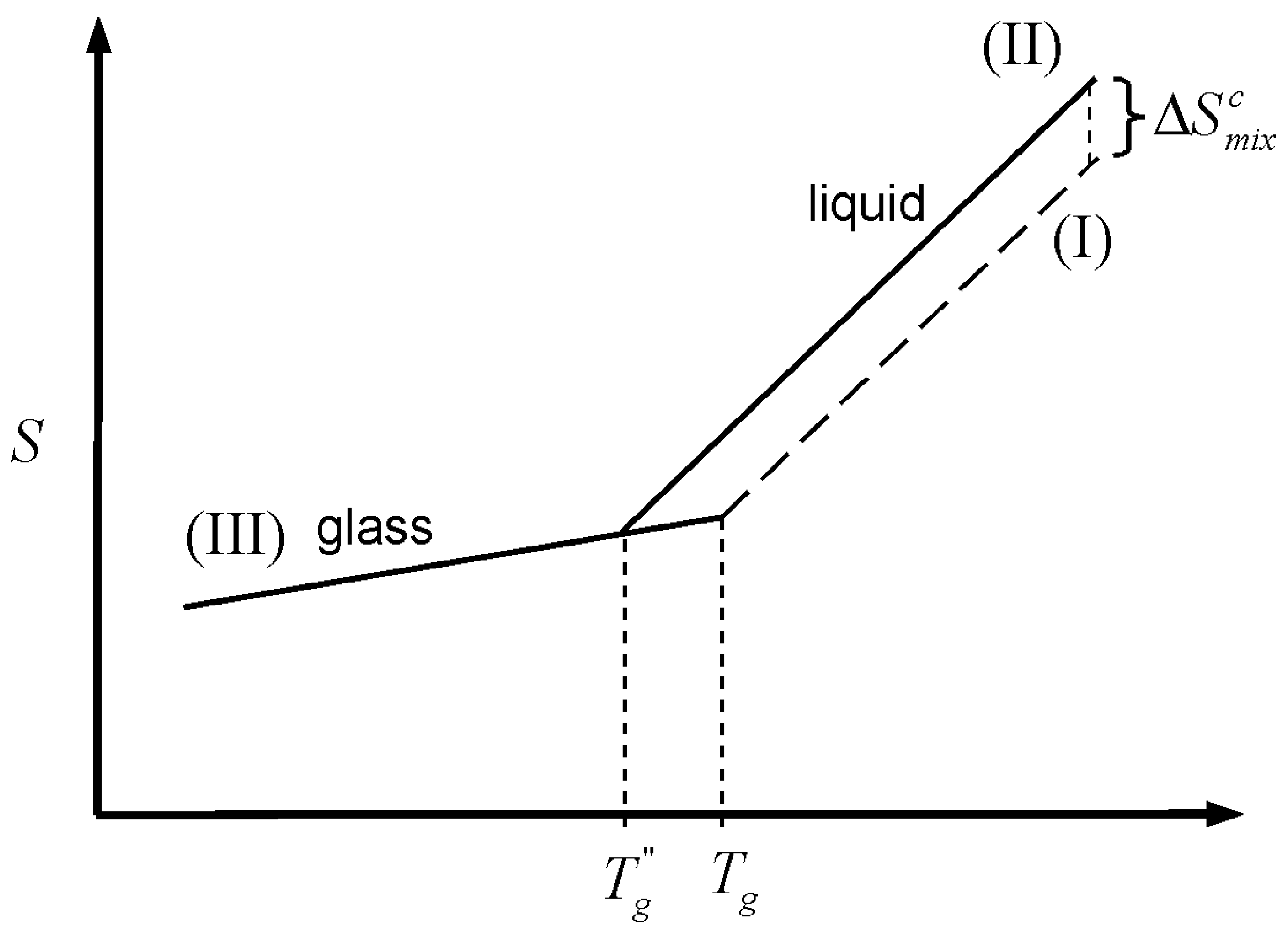 
For "remove barrier", the entropy change of each gas is the same as that of a gas expanding into a vacuum. Energy Unit Cost Steam Unit Cost Boiler Efficiency. 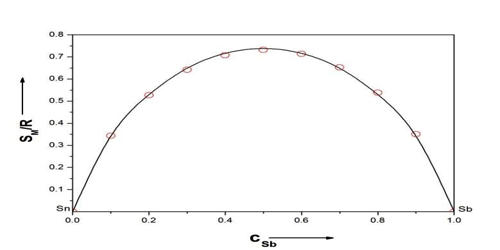
Click the play button next to "mix gases" to initiate mixing. Temperature drop (based on air ) Air (based on mixture temperature). In this Demonstration, ideal gases and are mixed isothermally by keeping the total volume constant (remove barrier option) or by adding gas to gas so the final volume is the same as the initial volume of (select "compress right"). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
